
- What is an AEM electrolyser, and how does it work?
- How does an AEM electrolyser differ from alkaline and PEM electrolysers in terms of membrane, electrolyte, catalysts, and operating philosophy?
- What are the key components of an AEM electrolyser stack, and what role does each component play?
- What materials are used for the membrane, electrodes, and catalysts in AEM electrolysers, and why are non-precious metals possible?
- What are the typical operating conditions of an AEM electrolyser (temperature, pressure, current density, electrolyte, water quality)?
- How is performance of an AEM electrolyser evaluated, and what factors limit efficiency and high-current-density operation?
- What are the main degradation and failure mechanisms in AEM electrolysers, and how do they affect lifetime?
- What safety considerations are critical in AEM electrolyser operation, particularly regarding hydrogen purity and gas crossover?
- How does the balance of plant (BoP) support AEM electrolyser operation, and which subsystems are most critical?
- What are the key advantages and current limitations of AEM electrolysers compared to other water electrolysis technologies?
- Where are AEM electrolysers best applied today, and how suitable are they for renewable and intermittent power integration?
- What are the main research directions and future developments needed for large-scale commercialization of AEM electrolysers?
What is an AEM electrolyser, and how does it work?
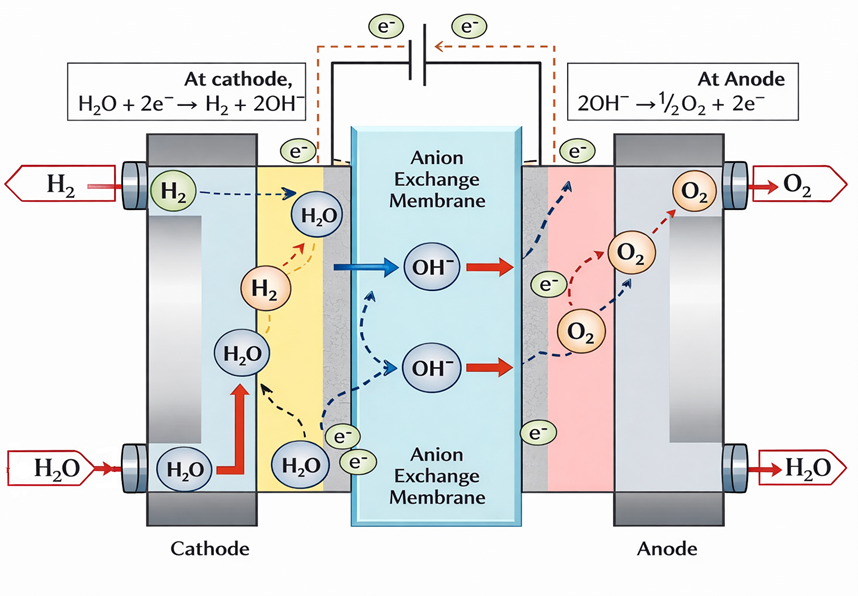
An Anion Exchange Membrane (AEM) electrolyser is a water electrolysis technology used to produce hydrogen gas. It splits water into hydrogen and oxygen using direct current (DC) electricity. Unlike proton exchange membrane electrolysers, an AEM electrolyser transports negatively charged hydroxide ions (OH⁻) through a solid polymer membrane. The membrane acts as both an ion conductor and a physical separator between hydrogen and oxygen gases. When DC power is supplied to the system, water is fed to the cathode side of the electrolyser. At the cathode, water molecules react with electrons to produce hydrogen gas and hydroxide ions. The hydrogen gas is collected at the cathode outlet. The hydroxide ions then move through the anion exchange membrane toward the anode due to the electric field. At the anode, the hydroxide ions undergo an oxidation reaction. This reaction produces oxygen gas, water, and releases electrons. The electrons flow through the external electrical circuit back to the cathode, completing the electrical loop. The membrane prevents direct mixing of hydrogen and oxygen while allowing ion transport.
This ion transport mechanism allows the electrolyser to operate in an alkaline chemical environment without the need for circulating concentrated liquid electrolytes. As a result, corrosion risks and maintenance requirements are reduced. The solid membrane structure also enables a compact and sealed cell design. Because the operating environment is alkaline, AEM electrolysers can use non-precious metal catalysts such as nickel-based materials. This helps reduce system cost compared to PEM electrolysers. Overall, AEM electrolysers combine alkaline electrochemistry with membrane-based design, offering a balance between efficiency, cost, safety, and system simplicity.
How does an AEM electrolyser differ from alkaline and PEM electrolysers in terms of membrane, electrolyte, catalysts, and operating philosophy?
AEM electrolysers sit between conventional alkaline electrolysers and proton exchange membrane (PEM) electrolysers. They combine features of both technologies in terms of chemistry and system design. Traditional alkaline electrolysers use a liquid electrolyte, usually potassium hydroxide (KOH), to transport hydroxide ions between the electrodes. This liquid electrolyte requires large tanks, piping, and circulation systems. As a result, alkaline electrolysers are bulky and have higher risks of corrosion and leakage. They also respond slowly to changes in electrical power, which limits their ability to operate with variable renewable energy sources.
PEM electrolysers use a solid polymer membrane instead of a liquid electrolyte. This membrane conducts protons in an acidic environment. Because of this design, PEM electrolysers are compact and can operate at high current densities. They also respond very quickly to changes in power. However, the acidic operating conditions require expensive materials. PEM systems use platinum-group metal catalysts and fluorinated membranes, which significantly increase system cost AEM electrolysers use a solid polymer membrane like PEM systems, but the membrane conducts hydroxide ions instead of protons. This allows the electrolyser to operate in an alkaline environment without circulating concentrated liquid electrolytes. As a result, the system design becomes simpler and more compact than traditional alkaline electrolysers. The alkaline environment also allows the use of non-precious metal catalysts, such as nickel-based materials. This helps reduce the overall cost of the system.
At the same time, the solid membrane structure allows AEM electrolysers to respond faster to changes in power compared to conventional alkaline systems. This makes them more suitable for operation with renewable energy sources such as solar and wind. Overall, AEM electrolysers are considered a hybrid technology. They aim to combine the low material cost of alkaline electrolysis with the compact design and operational flexibility of PEM electrolysers.
What are the key components of an AEM electrolyser stack, and what role does each component play?
An AEM electrolyser stack is made of many repeating electrochemical cells. Each cell works together to produce hydrogen efficiently. At the centre of each cell is the anion exchange membrane. This membrane allows hydroxide ions to pass through it. At the same time, it keeps hydrogen and oxygen gases separated. This separation is critical for safe operation. On both sides of the membrane, catalyst layers are present. The catalyst on the cathode side helps the hydrogen evolution reaction take place. The catalyst on the anode side supports the oxygen evolution reaction. These reactions would be very slow without catalysts. The catalyst layers are attached to porous electrodes or gas diffusion layers. These layers allow water to reach the reaction sites evenly. They also help the generated gases to escape easily. In addition, they provide good electrical conductivity between components.
Between each cell, bipolar plates are installed. These plates carry electrical current from one cell to the next. They also contain flow channels that guide water, hydrogen, and oxygen through the stack. Bipolar plates provide mechanical strength and help keep the cells properly aligned. Sealing components, such as gaskets, are used around the membrane and plates. These seals prevent leakage of gases and liquids. They also stop hydrogen and oxygen from mixing inside the stack. Proper sealing is essential for both efficiency and safety. All the cells are pressed together using a compression system. This system applies uniform pressure across the stack. Proper compression ensures good electrical contact between layers. It also maintains sealing integrity and prevents membrane damage. Consistent compression is essential for stable performance and long operating life of the electrolyser stack.
What materials are used for the membrane, electrodes, and catalysts in AEM electrolysers, and why are non-precious metals possible?
The materials used in AEM electrolysers are mainly influenced by their alkaline operating environment. This alkaline condition is created by the movement of hydroxide ions through the membrane. The anion exchange membrane is usually made from hydrocarbon-based polymers. These polymers contain positively charged functional groups that allow hydroxide ions to move through the membrane. Unlike PEM membranes, AEM membranes do not require fluorinated materials. This helps reduce material cost and lowers environmental impact. The alkaline environment also allows the use of non-precious metal catalysts. Nickel and nickel-based alloys are commonly used for both hydrogen and oxygen evolution reactions. These materials show good catalytic activity in alkaline conditions. They also remain stable during long-term operation. In acidic PEM systems, the same materials would corrode quickly and lose performance.
Other components, such as electrodes, flow plates, and structural parts, are also selected for alkaline compatibility. Stainless steel and nickel-coated steel are widely used for these components. These materials provide good corrosion resistance at a lower cost. In many cases, expensive materials like titanium are not required. The ability to use widely available and earth-abundant materials is a major advantage of AEM electrolyser technology. It significantly reduces capital cost and supports large-scale manufacturing. This cost advantage is one of the key reasons AEM electrolysers are considered promising for future green hydrogen production.
What are the typical operating conditions of an AEM electrolyser (temperature, pressure, current density, electrolyte, water quality)?
AEM electrolysers usually operate at moderate temperatures and pressures. These conditions are chosen to balance performance and material lifetime. Typical operating temperatures range from about 40 to 70 degrees Celsius. Higher temperatures improve reaction speed and ion conductivity in the membrane. However, very high temperatures can damage the membrane and catalysts over time. Operating pressure can vary depending on system design and application. Some AEM electrolysers operate close to atmospheric pressure. Others operate at pressures of several tens of bar. Higher operating pressure helps deliver hydrogen at useful pressure levels. This reduces the need for additional gas compression. At the same time, higher pressure increases mechanical stress on the membrane and other components. It also increases the risk of hydrogen and oxygen gas crossover.

Current density is another important operating parameter. Present AEM electrolysers typically operate in the range of 0.3 to 1.0 amperes per square centimetre. Higher current density increases hydrogen production from a given stack area. However, it also increases voltage losses and material stress. Ongoing research aims to increase current density without reducing system life. Most AEM electrolysers use deionized or high-purity water as the feed. In some systems, a very dilute alkaline electrolyte is added to improve ionic conductivity. Water purity is carefully controlled. Carbon dioxide must be kept to a minimum. CO₂ reacts with hydroxide ions to form carbonates. Carbonate formation reduces performance and accelerates degradation.
How is performance of an AEM electrolyser evaluated, and what factors limit efficiency and high-current-density operation?
The performance of an AEM electrolyser is measured using several key parameters. These include cell voltage, hydrogen production rate, energy efficiency, and Faradaic efficiency. Cell voltage is one of the most important indicators. For a given current density, a lower cell voltage means the electrolyser consumes less electrical energy. This directly improves overall efficiency. Different types of losses reduce the performance of an AEM electrolyser. Activation losses occur due to slow reaction kinetics at the electrodes. These losses are related to the activity of the catalysts. Ohmic losses occur due to electrical resistance in the membrane, electrodes, and contact interfaces. Higher resistance leads to higher voltage loss. Mass transport losses occur when water supply or gas removal is not uniform. These losses increase when hydrogen and oxygen cannot be removed quickly from the reaction sites.

Among all losses, membrane resistance and catalyst performance are especially important in AEM systems. A membrane with low ionic conductivity increases voltage loss. Poor catalyst activity increases activation overpotential. As current density increases, all losses become more significant. Ion transport through the membrane becomes more difficult. Gas bubbles also accumulate more easily at higher current densities. This increases voltage and limits high-power operation. To achieve high efficiency and stable performance, proper water management is essential. Water must reach the reaction sites evenly. Gas must be removed efficiently from the electrodes. Optimized electrode structures help improve mass transport. Improved membrane conductivity also helps reduce electrical losses. Together, these factors are critical for long-term and efficient AEM electrolyser operation.
What are the main degradation and failure mechanisms in AEM electrolysers, and how do they affect lifetime?
The long-term durability of AEM electrolysers depends on several connected degradation mechanisms. One of the main issues is membrane degradation. The anion exchange membrane can degrade chemically over time. This happens when hydroxide ions attack the functional groups inside the membrane. As a result, the membrane slowly loses its ability to conduct ions. Mechanical degradation of the membrane can also occur. The membrane can swell when it absorbs water and shrink when it dries. Repeated wet and dry cycles cause mechanical stress. Pressure differences between the anode and cathode sides also add stress. Over time, this can lead to cracks, thinning, or loss of mechanical strength.
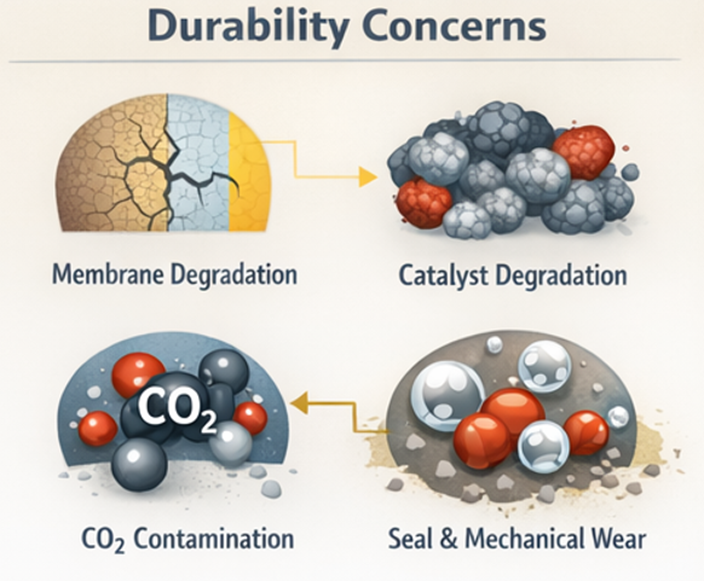
Catalyst degradation is another important factor affecting durability. Catalyst particles can agglomerate and grow in size during operation. This reduces the active surface area available for reactions. Catalysts can also oxidize or degrade due to repeated start-up and shutdown cycles. These redox cycles gradually reduce catalytic activity and increase cell voltage. Carbon dioxide contamination is a further challenge in AEM electrolysers. CO₂ from air can enter the system and react with hydroxide ions. This reaction forms carbonate species. Carbonates reduce hydroxide conductivity in the membrane. They can also block active sites on the catalysts and electrodes.
At the system level, mechanical and operational issues also affect lifetime. Seals and gaskets can degrade over time. Uneven compression across the stack can damage membranes and electrodes. Thermal cycling during operation can introduce additional stress. Together, these factors currently limit AEM electrolyser lifetimes to tens of thousands of operating hours. Ongoing improvements in materials and system design aim to extend operating life and improve long-term reliability.
What safety considerations are critical in AEM electrolyser operation, particularly regarding hydrogen purity and gas crossover?
Safety in AEM electrolyser operation mainly focuses on preventing hydrogen and oxygen from mixing. A mixture of these gases can be explosive. Safety systems also control pressure and temperature to keep the electrolyser within safe operating limits.
One of the main safety risks is gas crossover through the membrane. Gas crossover can occur if the membrane degrades over time. It can also happen if the pressure difference between the anode and cathode sides is not properly controlled. If hydrogen crosses into the oxygen side, or vice versa, the risk of ignition increases. To reduce this risk, AEM electrolysers use accurate pressure control systems. These systems maintain a small and safe pressure difference across the membrane. Gas purity is continuously monitored during operation. Sensors are used to detect hydrogen and oxygen leaks at an early stage.
Good sealing is also critical for safe operation. Gaskets and seals prevent gas leakage from the stack. Controlled start-up and shutdown procedures help avoid sudden pressure or temperature changes. Emergency venting systems are installed to safely release gases in abnormal situations. Although AEM electrolysers do not circulate concentrated alkaline electrolytes, safety remains a major design requirement. Hydrogen is highly flammable and reacts easily with oxygen. Therefore, AEM electrolysers still require careful safety design, monitoring, and operating procedures.
How does the balance of plant (BoP) support AEM electrolyser operation, and which subsystems are most critical?
The balance of plant, often called BoP, includes all supporting systems around the electrolyser stack. These systems are necessary for safe, stable, and efficient operation. The BoP does not produce hydrogen directly, but it enables the stack to operate properly. Power electronics are an important part of the BoP. Rectifiers convert alternating current from the grid or renewable sources into direct current. The electrolyser requires stable DC power to operate correctly. Power control also helps adjust current during start-up, shutdown, and load changes.
Water treatment systems supply high-purity water to the electrolyser. Impurities in water can damage the membrane and catalysts. Proper water treatment improves performance and extends system life. Cooling systems remove the heat generated during electrolysis. Temperature control is necessary to protect the membrane and maintain efficiency. The BoP also includes gas handling equipment. Gas–liquid separators remove water from hydrogen and oxygen streams. Dryers further reduce moisture content. Pressure control devices regulate gas pressure for safe storage or downstream use.
Instrumentation and control systems continuously monitor key operating parameters. These include voltage, current, temperature, pressure, and gas purity. The control system allows automatic operation and quick detection of faults. In many systems, the BoP represents a large part of the total system cost. It strongly influences overall efficiency, safety, and long-term reliability of the electrolyser.
What are the key advantages and current limitations of AEM electrolysers compared to other water electrolysis technologies?
AEM electrolysers offer several important advantages. One major advantage is their potential for lower cost. They can use non-precious metal catalysts instead of expensive platinum-group metals. They also use less costly structural materials compared to PEM electrolysers. This significantly reduces the overall capital cost of the system. AEM electrolysers use a solid membrane instead of a circulating liquid electrolyte. This removes the need to handle concentrated alkaline solutions. As a result, corrosion risk is lower and maintenance requirements are reduced. The system design becomes simpler and safer compared to traditional alkaline electrolysers.
Despite these advantages, AEM technology also has some limitations. Membrane durability is one of the main challenges. The membrane can degrade faster at high current densities. Long operating hours also increase the risk of performance loss. AEM electrolysers are sensitive to carbon dioxide contamination. CO₂ reacts with hydroxide ions and reduces membrane conductivity. This can lower efficiency and shorten system life. In addition, large-scale commercial deployment of AEM electrolysers is still limited. There are fewer long-term industrial references compared to alkaline and PEM technologies.
Because of these challenges, AEM electrolysers are currently considered an emerging technology. They show strong promise but are not yet a fully mature industrial solution. Continuous research and development are required to improve durability and enable wider adoption.
Where are AEM electrolysers best applied today, and how suitable are they for renewable and intermittent power integration?
At present, AEM electrolysers are mainly used for small to medium-scale hydrogen production. These applications are often sensitive to cost and require flexible operation. AEM electrolysers offer advantages in such cases due to their lower material cost and compact design. AEM electrolysers are well suited for integration with renewable energy sources. These include solar and wind power systems. Renewable power output often changes with time. AEM electrolysers can respond to these power fluctuations better than conventional alkaline electrolysers. This makes them suitable for variable and intermittent operation.
Common application areas include distributed hydrogen production systems. They are also used in pilot plants and industrial demonstration projects. These projects help validate performance and durability under real operating conditions. AEM electrolysers can handle intermittent operation reasonably well. However, they are still under development for highly dynamic operating conditions. At present, PEM electrolysers remain more robust for fast and frequent load changes. As AEM technology continues to improve, its reliability and operating range are expected to increase. In the future, AEM electrolysers are likely to be applied to larger-scale and more demanding hydrogen production applications.
What are the main research directions and future developments needed for large-scale commercialization of AEM electrolysers?
For AEM electrolysers to be widely commercialized, several improvements are still required. These improvements are needed in materials, system design, and manufacturing processes. One of the most important priorities is improving membrane stability. The membrane must resist chemical attack from hydroxide ions. It must also maintain mechanical strength over long operating periods. At present, membrane degradation is one of the main factors limiting system lifetime. Catalyst performance must also be improved. Catalysts need to remain active and stable at higher current densities. Higher current density allows more hydrogen to be produced from a smaller stack. This helps reduce system size and cost. Long-term catalyst durability is essential for continuous operation.
Another important area is tolerance to impurities. AEM electrolysers must better handle carbon dioxide from air. They must also tolerate small impurities in feed water. Improved impurity tolerance will simplify system design. It will also reduce operating and maintenance requirements. Manufacturing development is equally important. Production methods must be scalable and repeatable. This ensures consistent quality across large numbers of stacks. It also helps reduce manufacturing cost. With continued research and industrial investment, AEM electrolysers have strong potential. Over the next decade, they could become a key technology for producing cost-effective green hydrogen.

 Login
Login











